Post by Admin on Oct 3, 2022 19:46:49 GMT -7
Multi-Plume slab from the Woodward Ranch in Texas

Red plumes are usually iron, black is manganese.
The area from Marfa to the Woodward Ranch is blessed with marvelous Plumed Agates.
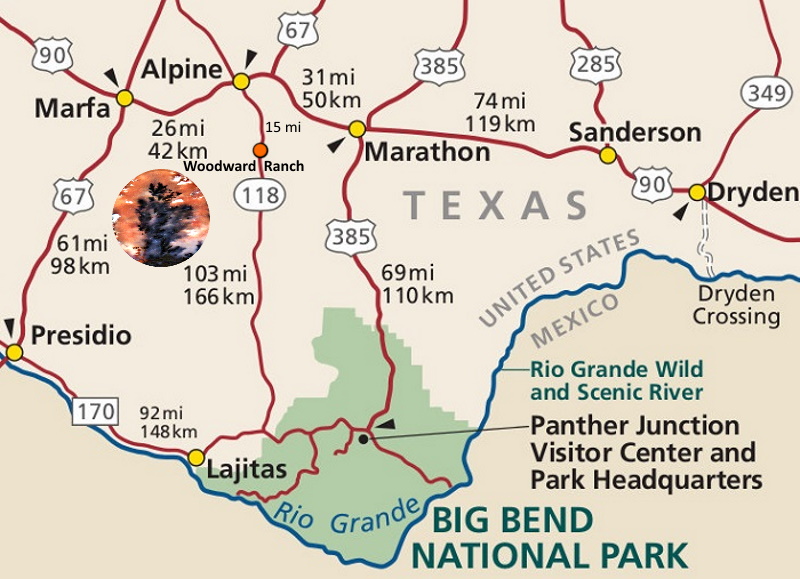
Moss, Ferns, Plumes, Dendrites ( tree like )
- How do those forms end up in rocks?
- How do those forms end up in rocks?
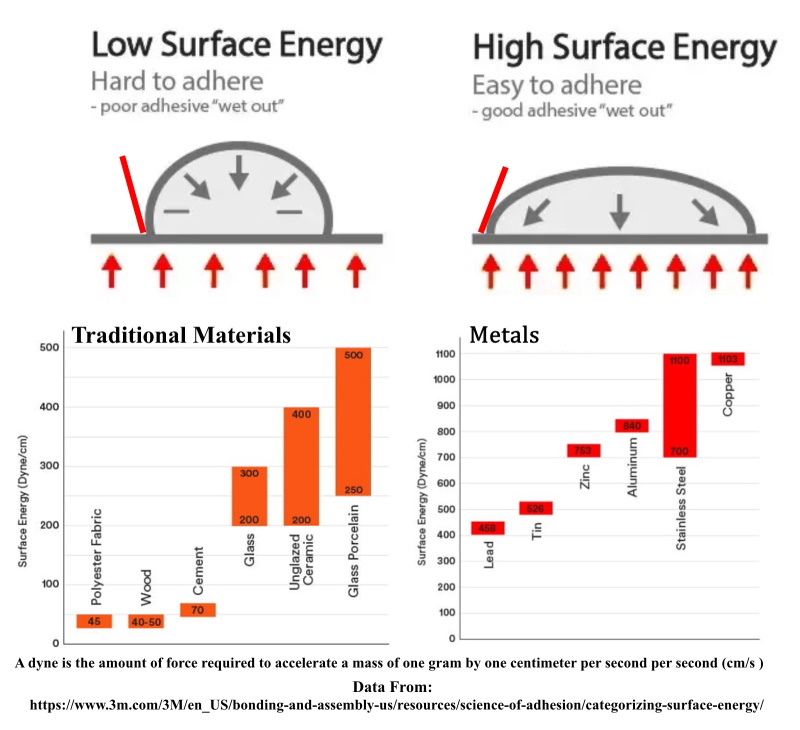
All matter is made of molecules. All molecules have varying degrees of attraction or repulsion to or from other molecules. That intermolecular attraction is why the molecules of liquids like water, or even solids like ice, stay together in a drop or puddle rather than drift apart into the air. The attractive force of the molecules present at the surface of a liquid towards each other is called the surface tension of that liquid.
Surface energy is the equivalent attractive force present between the molecules at the surface of a solid substance. This attractive force is what holds a substance together into a cohesive form. We usually use surface tension when referring to liquids and surface energy when referring to solids, but in reality they are the same thing.
Surface Tension is usually measured in newtons per meter. Represented by the symbol γ (alternatively σ or T), is measured in force per unit length. Its SI unit is newton per meter but the cgs unit of dyne per centimeter is also used.
Surface Energy, measured in DYNES - a dyne is the amount of force required to accelerate a mass of one gram by one centimeter per second per second (cm/s )- may be defined as the excess energy at the surface of a material compared to the bulk, or it is the work required to build an area of a particular surface. Another way to view the surface energy is to relate it to the work required to cut a bulk sample, creating two surfaces. There is "excess energy" as a result of the now-incomplete, unrealized bonding at the two surfaces.
Once nucleation has occurred, solidification proceeds by the movement of an interface. The process may generate heat if the enthalpy of the solid is less than that of the liquid. Similarly, solute may partition into the liquid if its solubility in the solid is less than that in the liquid.
Ground Water
Water is a solvent and dissolves minerals from the rocks with which it comes in contact. Ground water may contain dissolved minerals and gases that give it the tangy taste enjoyed by many people. Without these minerals and gases, the water would taste flat.
A wide range of different elements can become dissolved in groundwater as a result of interactions with the atmosphere, the surficial environment, soil and bedrock. Groundwaters tend to have much higher concentrations of most constituents than do surface waters, and deep groundwaters that have been in contact with rock for a long time tend to have higher concentrations than shallow and or young waters.
The most common dissolved mineral substances are cations of sodium, calcium, magnesium, potassium, iron, and anions chloride, bicarbonate, and sulfate. In water chemistry, these substances are called common constituents.
A test of the acidity of water is pH, - a measure of the hydrogen-ion concentration. The pH scale ranges from 0 to 14. A pH of 7 indicates neutral water; greater than 7, the water is basic; less than 7, it is acidic. A one unit change in pH represents a 10-fold difference in hydrogen-ion concentration. For example, water with a pH of 6 has 10 times more hydrogen-ions than water with a pH of 7. Water that is basic can form scale in pipes; acidic water can corrode pipes. According to U.S. Environmental Protection Agency criteria, water for domestic use should have a pH between 5.5 and 9.
Sodium Water with less than a thousand mg/L (milligrams per liter) of dissolved minerals is classed as slightly saline. If the quantity of dissolved minerals exceeds 1,000 mg/L cannot be tolerated by humans, animals, or plants. Many parts of the Nation are underlain at depth by highly saline ground water that has only very limited uses without being purified.
Water that contains a lot of calcium and magnesium is said to be hard. The hardness of water is expressed in terms of the amount of calcium carbonate-the principal constituent of limestone-or equivalent minerals that would be formed if the water were evaporated. Water is considered soft if it contains 0 to 60 mg/L of hardness, moderately hard from 61 to 120 mg/L, hard between 121 and 180 mg/L, and very hard if more than 180 mg/L. Very hard water is not desirable for many domestic uses; it will leave a scaly deposit on the inside of pipes, boilers, and tanks. Hard water can be softened at a fairly reasonable cost, but it is not always desirable to remove all the minerals that make water hard. Extremely soft water is likely to corrode metals.
Ground water, especially if the water is acidic, in many places contains excessive amounts of iron. Iron causes reddish stains. Like hardness, excessive iron content can be reduced by treatment.
Ground water in earthquake prone areas will briefly contain large amounts of silica jells.
web.viu.ca/earle/geol304/304g.pdf
(The Nanaimo Group is a geologic group in both British Columbia and Washington state. It preserves fossils dating back to the Cretaceous period.)
Bonding Energy
- www.3m.com/3M/en_US/bonding-and-assembly-us/resources/science-of-adhesion/categorizing-surface-energy/
The accumulation of solute and heat ahead of the interface can lead to circumstances in which the liquid in front of the solidification front is supercooled. The interface thus becomes unstable and in appropriate circumstances solidification becomes dendritic.
A dendrite tends to branch because the interface instability applies at all points along its growth front. The branching gives it a tree-like character which is the origin of the term dendrite.
Most metals and alloys crystallize in one of three very common structures: body-centered cubic (bcc), cubic close packed (ccp, also called face centered cubic, fcc), or hexagonal close packed (hcp).
Growth tends to occur along fast growth directions which are generally <100> for cubic metals
msestudent.com/miller-indices/
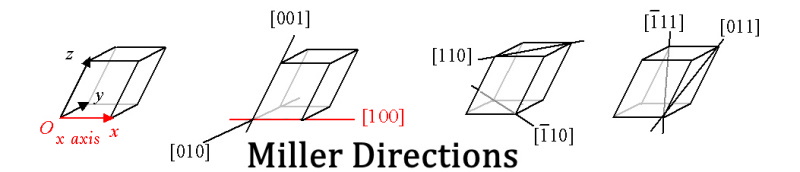
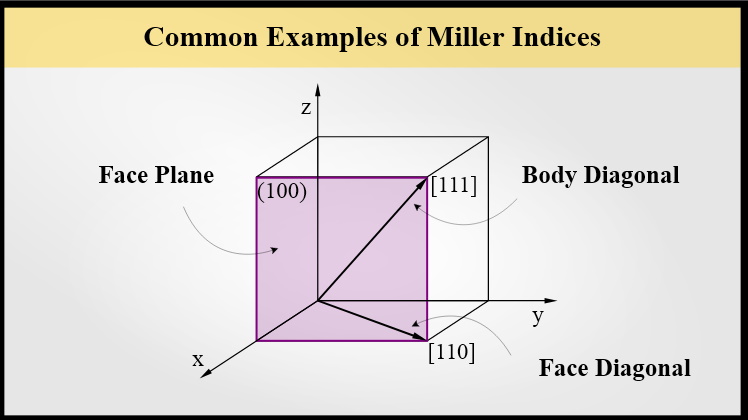
Crystals are defined by their symmetry. That’s why the choice of origin is arbitrary. In some cases, we may want to distinguish between a specific direction, and all equivalent directions. We make this distinction with brackets.
Square brackets [] indicate a specific direction. For example, in a cubic system [100] and [010] are perpendicular directions.
Angle brackets <> indicate a family of directions. For example, in a cubic system \langle 100 \rangle includes these directions [100], [\bar{1}00], [010], [0\bar{1}0], [001], and [00\bar{1}].
Parenthesis () indicate a specific plane. For example, in a cubic system (100) and (010) are perpendicular planes.
Curly brackets {} indicate a family of planes. For example, in a cubic system \{100\} includes (100), (\bar{1}00), (010), (0\bar{1}0), (001), and (00\bar{1}).
Square brackets [] indicate a specific direction. For example, in a cubic system [100] and [010] are perpendicular directions.
Angle brackets <> indicate a family of directions. For example, in a cubic system \langle 100 \rangle includes these directions [100], [\bar{1}00], [010], [0\bar{1}0], [001], and [00\bar{1}].
Parenthesis () indicate a specific plane. For example, in a cubic system (100) and (010) are perpendicular planes.
Curly brackets {} indicate a family of planes. For example, in a cubic system \{100\} includes (100), (\bar{1}00), (010), (0\bar{1}0), (001), and (00\bar{1}).

www.mrl.ucsb.edu/~seshadri/2017_100A/GuideToPointsDirectionsPlanes.pdf
msestudent.com/miller-indices/

www.phase-trans.msm.cam.ac.uk/dendrites.html
Battery Fires
Professor Clare Grey and her team at Cambridge University have concluded that metal fibres in the form of dendrites grow within lithium batteries which are charged rapidly. These cause short circuits, overheating and in some cases, fires.



