Post by Admin on Jan 2, 2023 18:16:41 GMT -7
Copper is a pure single element. The crystal system of natural copper is cubic. It has a metallic luster and a specic gravity of 8-9 with a hardness of 2-1/2 to 3 and can be easily scratched with a knife. Nautal copper has no cleavage and its fracture is hackley. This element is heavy, ductile and malleable. Native copper is copper red on fresh fracture but may be greenish or bluish or tarnished if weathered. It is often found with small amounts of arsenic, antimony, bismuth, iron, silver, and gold.
Copper only comes into existence late in a star's life.

Only after iron burning begins in stars does copper have a chance of coming into existence.
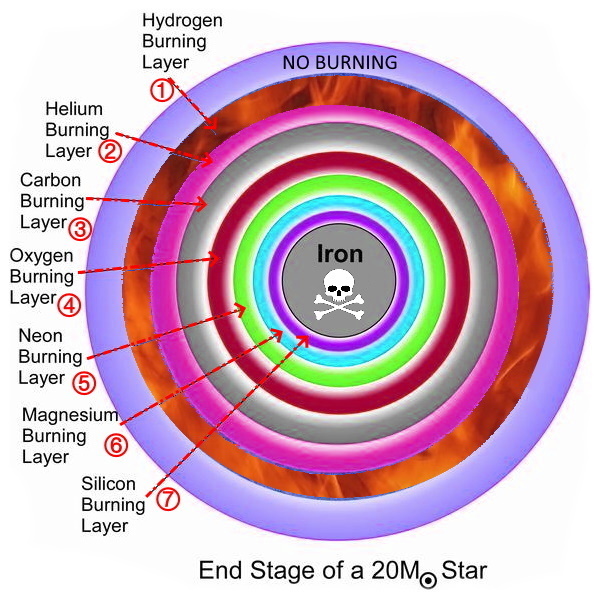
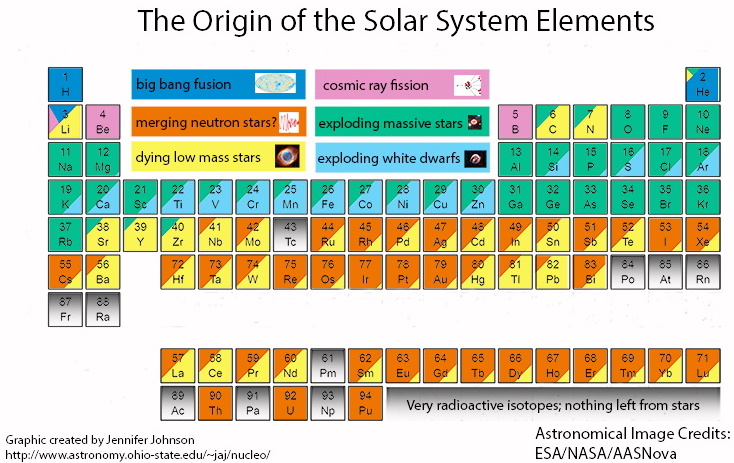
Beginning with iron, the elements use more energy than they create, spelling the doom of the star. Processes as the star collapses, goes Supernova, do the heavier metals form as Helium atoms are forced into #26 iron, making it 27, then 28 and 29 copper.

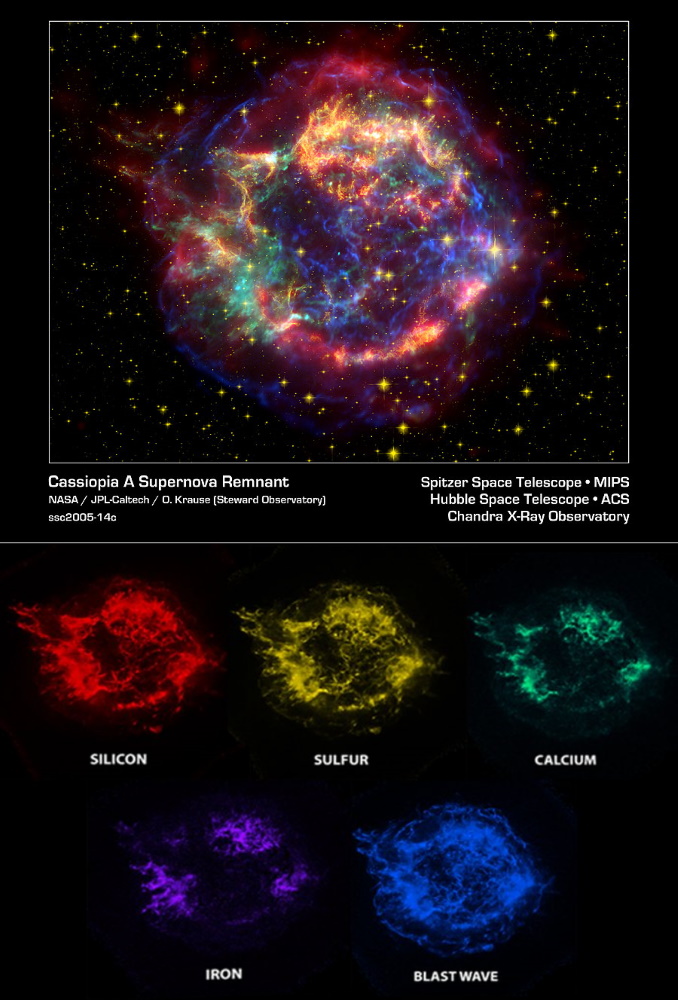
It is blasted through space with it's surrounding elements, mixing with the rest of the star debris where the copper begins colliding with itself and other elements like oxygen and sulfur.
[/div]
Copper is a Transition Metal, having in addition to their outer-shell electrons, can involve their inner-shell electrons in chemical bonding. The inner d-subshell electrons of transition metals are also called valence electrons, so, the valence electrons of a transition metal can be determined using its electronic configuration, by taking the sum of its ns and (n-1)d electrons.
First-row transition metals have a similar electronic configuration that ends in 3d1-10 4s2, but 24 Chromium and 29 Copper are exceptions with a half-filled 4s1 atomic orbital.
Copper only comes into existence late in a star's life.

Only after iron burning begins in stars does copper have a chance of coming into existence.


Beginning with iron, the elements use more energy than they create, spelling the doom of the star. Processes as the star collapses, goes Supernova, do the heavier metals form as Helium atoms are forced into #26 iron, making it 27, then 28 and 29 copper.

It is blasted through space with it's surrounding elements, mixing with the rest of the star debris where the copper begins colliding with itself and other elements like oxygen and sulfur.

[/div]
Millions of years later the copper elements enter nebulae where they collide with even more other elements.
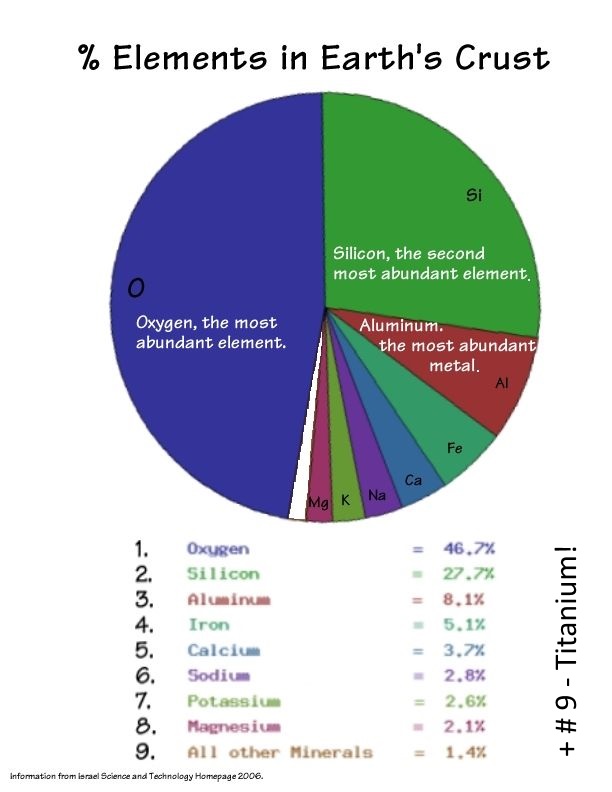
Finally copper, as dust, meteors, asteroids, and comets collide with earth where it first encounters lots of oxygen, then silicon, aluminum, iron, calcium, sodium, and the rest of our crust materials.

Copper is a Transition Metal, having in addition to their outer-shell electrons, can involve their inner-shell electrons in chemical bonding. The inner d-subshell electrons of transition metals are also called valence electrons, so, the valence electrons of a transition metal can be determined using its electronic configuration, by taking the sum of its ns and (n-1)d electrons.
First-row transition metals have a similar electronic configuration that ends in 3d1-10 4s2, but 24 Chromium and 29 Copper are exceptions with a half-filled 4s1 atomic orbital.
Copper Ores
Copper(I) oxide (cuprous oxide, Cu2O)
Copper(II) oxide (cupric oxide, CuO)
Copper peroxide (CuO2)
Copper(III) oxide (Cu2O3)
Copper(IV) oxide (CuO2)[1]
silicated chrysocolla (chalcedony stained with copper salts) and some do contain a fair am amount of quartz or other hard minerals. Although most commonly the main matrix of copper ores are dolomote and limonite.
Malachite (pronounced mala-kite) is usually a bright green color and has a nonmetallic luster. It has a light green streak and can always be scratched with a knife. Malachite, a copper carbonate, is an important ore of copper and is a good indicator of copper deposits. In its pure form it contains 57% copper, the rest is made up of carbonate and water.
Azurite also is a copper carbonate. It's streak is light blue. Malachite and azurite frequently occur together and are found in the upper weathered (oxidized) zones of copper ore bodies. Azurite is the scarcer of the two has a soft blue color.
Chalcopyrite is an iron-copper sulfide. It has a brass yellow color. It is distinguished from pyrite by being softer and yellower. Its golden glint when in small specks in quartz often is mistaken for gold. The glint will disappear when turned at certain angles to the light while gold appears the same at all angles.
Chalcopyrite is the primary ore of copper and is prevalent wherever copper ore is being mined below the surface zone.
Chalcocite is a copper sulfate. It is one of the highest grade and most important ores of copper and is opaque with a dark lead gray to black color.
Chalcocite is often associated with and shows alteration to azurite, bornite, covellite, malachite, and native copper. Important deposits are found in Arizona's Bagdad, Jerome, and Superior areas. Other localities include Bingham, Utah; Santa Rita, New Mexico; Ely, Nevada, and the Genesee Valley district in California.
Bornite is a copper-iron sulfate. Its color is a natural bronze, but on exposure it tarnishes to the varigated colors that have caused it to be nicknamed ìPeacock ore.î It is rarely found on the surface but is prevalent in deeper levels of copper mines.
Turquoise is a hydrous aluminum phosphate with copper. To be desirable for gems the color should be green blue. The color is due to the presence of copper and is found near the surface of copper deposits. Sometimes in may appear as an outcrop.
Chrysocolla has various shades of blue to green and is a hydrous copper silicate. It is often found with azurite and malachite. Although its color is attractive, it is too soft to make good gem stones. Be aware of this fact when buying jewelry. Sometimes chrysocolla is passed off as turquoise.
Copper(I) oxide (cuprous oxide, Cu2O)
Copper(II) oxide (cupric oxide, CuO)
Copper peroxide (CuO2)
Copper(III) oxide (Cu2O3)
Copper(IV) oxide (CuO2)[1]
silicated chrysocolla (chalcedony stained with copper salts) and some do contain a fair am amount of quartz or other hard minerals. Although most commonly the main matrix of copper ores are dolomote and limonite.
Malachite (pronounced mala-kite) is usually a bright green color and has a nonmetallic luster. It has a light green streak and can always be scratched with a knife. Malachite, a copper carbonate, is an important ore of copper and is a good indicator of copper deposits. In its pure form it contains 57% copper, the rest is made up of carbonate and water.
Azurite also is a copper carbonate. It's streak is light blue. Malachite and azurite frequently occur together and are found in the upper weathered (oxidized) zones of copper ore bodies. Azurite is the scarcer of the two has a soft blue color.
Chalcopyrite is an iron-copper sulfide. It has a brass yellow color. It is distinguished from pyrite by being softer and yellower. Its golden glint when in small specks in quartz often is mistaken for gold. The glint will disappear when turned at certain angles to the light while gold appears the same at all angles.
Chalcopyrite is the primary ore of copper and is prevalent wherever copper ore is being mined below the surface zone.
Chalcocite is a copper sulfate. It is one of the highest grade and most important ores of copper and is opaque with a dark lead gray to black color.
Chalcocite is often associated with and shows alteration to azurite, bornite, covellite, malachite, and native copper. Important deposits are found in Arizona's Bagdad, Jerome, and Superior areas. Other localities include Bingham, Utah; Santa Rita, New Mexico; Ely, Nevada, and the Genesee Valley district in California.
Bornite is a copper-iron sulfate. Its color is a natural bronze, but on exposure it tarnishes to the varigated colors that have caused it to be nicknamed ìPeacock ore.î It is rarely found on the surface but is prevalent in deeper levels of copper mines.
Turquoise is a hydrous aluminum phosphate with copper. To be desirable for gems the color should be green blue. The color is due to the presence of copper and is found near the surface of copper deposits. Sometimes in may appear as an outcrop.
Chrysocolla has various shades of blue to green and is a hydrous copper silicate. It is often found with azurite and malachite. Although its color is attractive, it is too soft to make good gem stones. Be aware of this fact when buying jewelry. Sometimes chrysocolla is passed off as turquoise.
