Post by 1dave on Sept 6, 2020 15:59:35 GMT -7
Basic Info: Water flows through the crust dissolving calcium and magnesium.
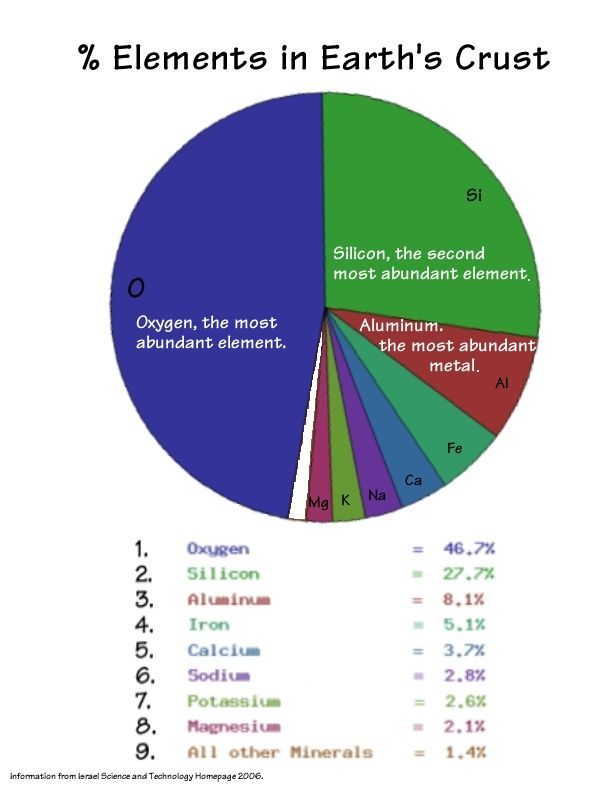
Each mineral ion has a charge slightly different from others making it possible to design specific charges in batteries.


Part one - water softeners
Extracted from: www.popularmechanics.com/home/interior-projects/how-to/a150/1275126/
link
Water is a tool that carries material from one place to the next by holding things - floating, suspending, or dissolving them.
An electrician friend used a bucket and an irrigation ditch to drag miles of heavy electrical cable in place!
The last item is why the dishes you thought were washed are covered with spots when dry, why the water in your shower leaves a film on everything it touches, and why what you thought was clean water has clogged up your plumbing system.
The Problem is The Solution
Ground water picks up soluble bits of whatever it passes through. This can mean contamination making the water unfit to drink, but usually it simply means that the water contains minerals found in the earth. Of these, calcium and magnesium make our water hard. That causes soaps and detergents to combine with the minerals to form a coagulated soap curd that hangs around clinging to everything. This can be a big problem building up inside your plumbing!
The solution to the problem is to get rid of the calcium and magnesium. While there are chemical treatments that do this, the most popular answer is a water softener. All water softeners use the same operating principle: They trade the minerals for something else, in most cases sodium. The process is called ion exchange.

The heart of a water softener is a mineral tank. It's filled with small polystyrene beads, also known as resin or zeolite. The beads carry a negative charge. Both calcium and magnesium in water carry positive charges that cause them to cling to the beads as hard water passes through the mineral tank..
Water softeners have a separate brine tank that uses common salt to create a brine solution.
You also can use potassium chloride instead of salt (sodium chloride), although this costs about three to four times more.
Sodium and potassium ions also have positive charges, but not as strong as the charge on the calcium and magnesium. When a very strong brine solution is flushed through a tank that has beads already saturated with calcium and magnesium, the sheer volume of the sodium ions is enough to drive the calcium and magnesium ions off the beads.

In normal operation, hard water moves into the mineral tank and the calcium and magnesium ions move to the beads, replacing sodium ions. The sodium ions go into the water. Once the beads are saturated with calcium and magnesium, the unit enters a 3-phase regenerating cycle.

1. The backwash phase reverses water flow to flush dirt out of the tank.
2. In the recharge phase, the concentrated sodium-rich salt solution is carried from the brine tank through the mineral tank. The sodium collects on the beads, replacing the calcium and magnesium, which go down the drain.
3. The mineral tank is flushed of excess brine and the brine tank is refilled.
Most popular water softeners have an automatic regenerating system.
The most basic type has an electric timer that flushes and recharges the system on a regular schedule. During recharging, soft water is not available.
A second type of control uses a computer that watches how much water is used. When enough water has passed through the mineral tank to have depleted the beads of sodium, the computer triggers regeneration. These softeners often have reserve resin capacity, so that some soft water will be available during recharging.
A third type of control uses a mechanical water meter to measure water usage and initiate recharging. The advantage of this system is that no electrical components are required and the mineral tank is only recharged when necessary. When it is equipped with two mineral tanks, softened water is always available, even when the unit is recharging.
Judging Water Hardness
Companies that sell water softening equipment generally offer test kits that help you determine the hardness of your water. For commercial testing sources, check your Yellow Pages under "water analysis."
Water hardness is measured in grains per gallon (GPG) or milligrams per liter (mg/l, equivalent to parts per million, or ppm). Water up to 1 GPG (or 17.1 mg/l) is considered soft, and water from 60 to 120 GPG is considered moderately hard. A water softener's effectiveness depends on how hard the incoming water is. Water over 100 GPG may not be completely softened.
Health Concerns:
Normal hard water poses no health hazard. On the other hand, the sodium that remains in softened water may be a problem for those on sodium-restricted diets. Other people simply may wish to avoid the slightly salty taste of treated water. In either case you can install a separate water dispenser that bypasses the softener.
Part Two - using Magnetism:
Magnetic Water/Fluid Conditioners have been around for much longer than most people are aware. They have now been used worldwide for over a century in both residential and industrial applications.
There are at least 80 patents filed with the US Patent Office just for "Permanent Magnet Fluid Conditioners". One of the first patents on this science was US Patent No. 531,183*, which was filed October 17, 1890 and was granted Dec 18, 1894. One of the newest patents granted in this area is US Patent No. 6,716,346* which was granted April 4, 2004.
These devices have been in use now for 115 years and there are still new patents being applied for.
There is a whole other group of patents for these devices that use electro-magnetics in place of permanent magnets.
Do they work? Lots of "scientists" who have never experimented with them say no!
People who have tried them say yes and no. Who knows what they did.
Finally a Real scientist in Ireland did a Real Experiment.
This is the results:
The top chart shows how long the crystallization lasted during the time (200 hours!)of the experiment.
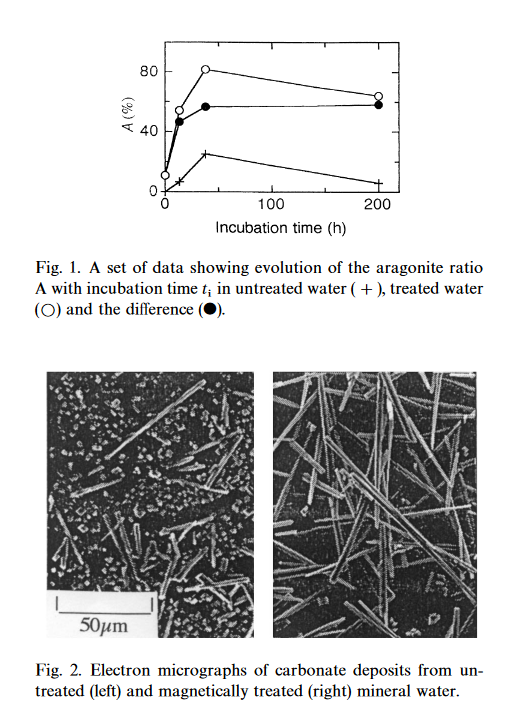
As water passes through a strong magnetic field, calcite scales crystallize into aragonite rods!
They don't stick to your plumbing or soap suds.
Magnesium remains to be experimented with.

Each mineral ion has a charge slightly different from others making it possible to design specific charges in batteries.


Part one - water softeners
Extracted from: www.popularmechanics.com/home/interior-projects/how-to/a150/1275126/
link
Water is a tool that carries material from one place to the next by holding things - floating, suspending, or dissolving them.
An electrician friend used a bucket and an irrigation ditch to drag miles of heavy electrical cable in place!
The last item is why the dishes you thought were washed are covered with spots when dry, why the water in your shower leaves a film on everything it touches, and why what you thought was clean water has clogged up your plumbing system.
The Problem is The Solution
Ground water picks up soluble bits of whatever it passes through. This can mean contamination making the water unfit to drink, but usually it simply means that the water contains minerals found in the earth. Of these, calcium and magnesium make our water hard. That causes soaps and detergents to combine with the minerals to form a coagulated soap curd that hangs around clinging to everything. This can be a big problem building up inside your plumbing!
The solution to the problem is to get rid of the calcium and magnesium. While there are chemical treatments that do this, the most popular answer is a water softener. All water softeners use the same operating principle: They trade the minerals for something else, in most cases sodium. The process is called ion exchange.

The heart of a water softener is a mineral tank. It's filled with small polystyrene beads, also known as resin or zeolite. The beads carry a negative charge. Both calcium and magnesium in water carry positive charges that cause them to cling to the beads as hard water passes through the mineral tank..
Water softeners have a separate brine tank that uses common salt to create a brine solution.
You also can use potassium chloride instead of salt (sodium chloride), although this costs about three to four times more.
Sodium and potassium ions also have positive charges, but not as strong as the charge on the calcium and magnesium. When a very strong brine solution is flushed through a tank that has beads already saturated with calcium and magnesium, the sheer volume of the sodium ions is enough to drive the calcium and magnesium ions off the beads.

In normal operation, hard water moves into the mineral tank and the calcium and magnesium ions move to the beads, replacing sodium ions. The sodium ions go into the water. Once the beads are saturated with calcium and magnesium, the unit enters a 3-phase regenerating cycle.

1. The backwash phase reverses water flow to flush dirt out of the tank.
2. In the recharge phase, the concentrated sodium-rich salt solution is carried from the brine tank through the mineral tank. The sodium collects on the beads, replacing the calcium and magnesium, which go down the drain.
3. The mineral tank is flushed of excess brine and the brine tank is refilled.
Most popular water softeners have an automatic regenerating system.
The most basic type has an electric timer that flushes and recharges the system on a regular schedule. During recharging, soft water is not available.
A second type of control uses a computer that watches how much water is used. When enough water has passed through the mineral tank to have depleted the beads of sodium, the computer triggers regeneration. These softeners often have reserve resin capacity, so that some soft water will be available during recharging.
A third type of control uses a mechanical water meter to measure water usage and initiate recharging. The advantage of this system is that no electrical components are required and the mineral tank is only recharged when necessary. When it is equipped with two mineral tanks, softened water is always available, even when the unit is recharging.
Judging Water Hardness
Companies that sell water softening equipment generally offer test kits that help you determine the hardness of your water. For commercial testing sources, check your Yellow Pages under "water analysis."
Water hardness is measured in grains per gallon (GPG) or milligrams per liter (mg/l, equivalent to parts per million, or ppm). Water up to 1 GPG (or 17.1 mg/l) is considered soft, and water from 60 to 120 GPG is considered moderately hard. A water softener's effectiveness depends on how hard the incoming water is. Water over 100 GPG may not be completely softened.
Health Concerns:
Normal hard water poses no health hazard. On the other hand, the sodium that remains in softened water may be a problem for those on sodium-restricted diets. Other people simply may wish to avoid the slightly salty taste of treated water. In either case you can install a separate water dispenser that bypasses the softener.
Part Two - using Magnetism:
Magnetic Water/Fluid Conditioners have been around for much longer than most people are aware. They have now been used worldwide for over a century in both residential and industrial applications.
There are at least 80 patents filed with the US Patent Office just for "Permanent Magnet Fluid Conditioners". One of the first patents on this science was US Patent No. 531,183*, which was filed October 17, 1890 and was granted Dec 18, 1894. One of the newest patents granted in this area is US Patent No. 6,716,346* which was granted April 4, 2004.
These devices have been in use now for 115 years and there are still new patents being applied for.
There is a whole other group of patents for these devices that use electro-magnetics in place of permanent magnets.
Do they work? Lots of "scientists" who have never experimented with them say no!
People who have tried them say yes and no. Who knows what they did.
Finally a Real scientist in Ireland did a Real Experiment.
This is the results:
The top chart shows how long the crystallization lasted during the time (200 hours!)of the experiment.

As water passes through a strong magnetic field, calcite scales crystallize into aragonite rods!
They don't stick to your plumbing or soap suds.
Magnesium remains to be experimented with.
