Post by 1dave on Nov 7, 2020 18:29:13 GMT -7
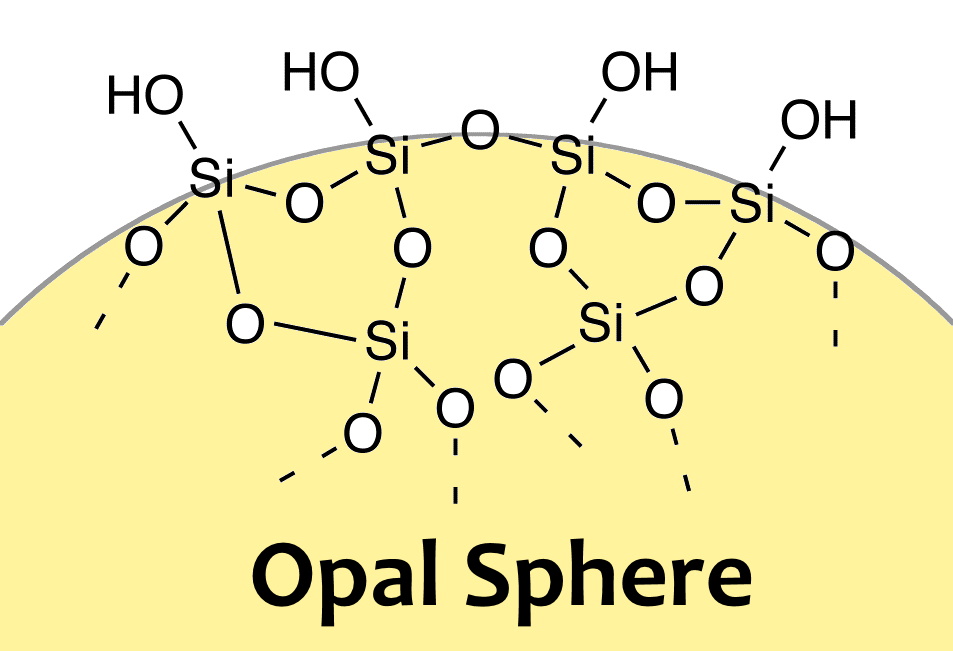
Silicon atoms NEED 4 electrons to satiate its electromagnetic needs. Only half that need is solved in the silica molecule, so SiO2 is a "hungry molecule" - Si wanting 2 more electrons, and the O each having an electron to spare, wanting and offering - Being both a Giver and a Taker at the same time.
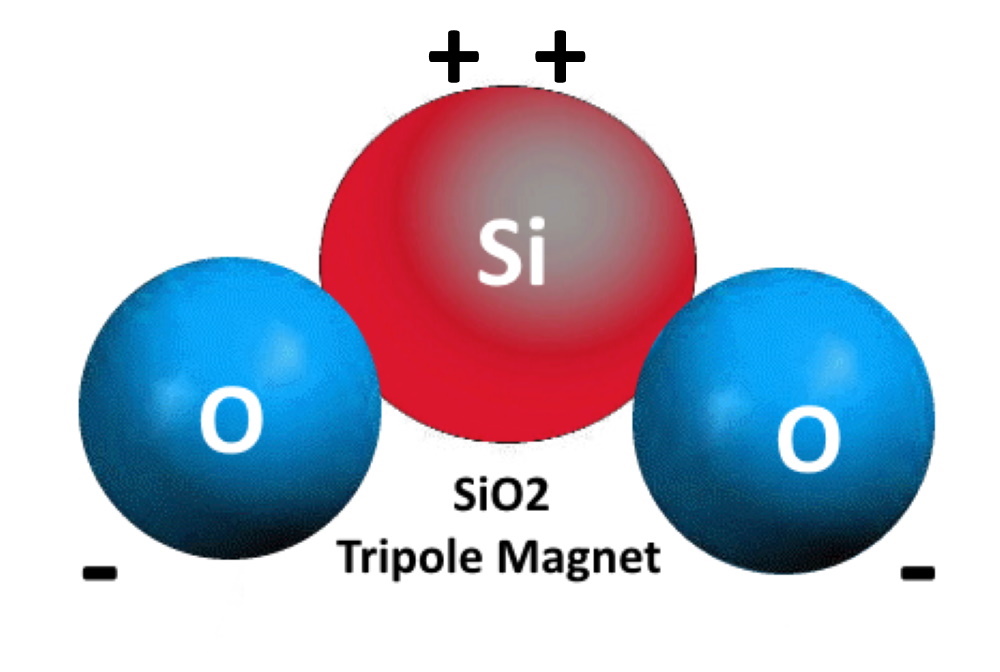

A word about electricity and magnetism. They are opposite sides of the same coin. You can't have one without the other.
Electro-Magnetism! - - - - - - - - - - -
As the most common mineral in earth's crust, it usually combines with other SiO2 molecules to form SiO4 tetrahedrons, which then join other tetrahedrons - becoming ever larger Givers and Takers!.


ALL substances go through a series of changes with variations of heat and pressure.


Here is a diagram for SiO2 -

The letters pH stand for potential of Hydrogen. The pH scale was devised in 1923 by Danish biochemist Søren Peter Lauritz Sørensen (1868-1969). In effect pH is a measure of the concentration of hydrogen ions (that is, protons) in a substance.
pH is a measure of how acidic/basic water is. The range goes from 0 - 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base. Again, pH is just a measure of the relative amount of free hydrogen and hydroxyl ions in the water.

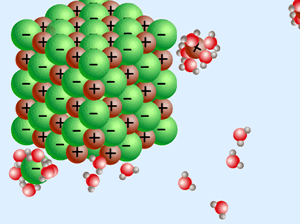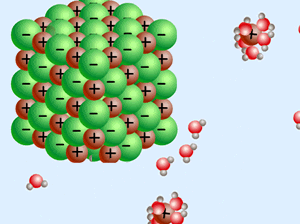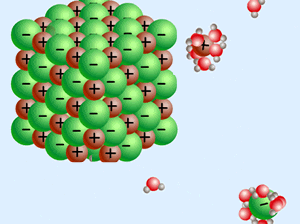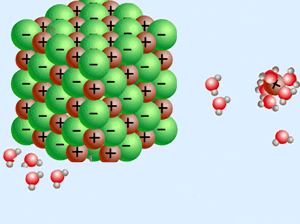
Some salts dissolve easier in acids, others in bases.
Silica Jells are amorphous and porous forms of silicon dioxide, consisting of an irregular tridimensional framework of alternating silicon and oxygen atoms with nanometer-scale voids and pores. The voids may contain water or some other liquids, or may be filled by gas or vacuum.
Opal-A has a solubility of 120–140 ppm in normal marine sediment pore water, cristobalite25–30 ppm, and quartz 6–10 ppm. The dissolution of opal-A generally results in supersaturation with respect to opal-CT, which is precipitated in preference to quartz.
Opal-CT consists of small bladed crystals that form spherical lepispheres 5–10mm in diameter. (Lepisphere : A microcrystalline, bladeshaped crystal of a metastable variety of quartz, composed of cristobalite with interlayered lattices of tridymite, aggregates of which often occur during the transformation of opal into quartz chert.)
Opal-C consists of small bladed cristobalite crystals that form spherical lepispheres 5–10mm in diameter.
Quartz The solubility in water of both amorphous silica and quartz increases rapidly above pH 9, and thus a sharp reduction in alkalinity from pH>9 results in silica precipitation. Precipitation generally takes place from dilute solutions over longer periods of time. The precipitation of silica, particularly around hot springs, occurs due to the evaporation of silica-saturated water. Silica solubility is higher in hot water, and thus cooling is a trigger for silica deposition. Boiling is also a trigger for silica precipitation in hydrothermal systems.
Glass is a non-crystalline, often transparent amorphous solid typically transparent or translucent, created by meteor impacts, volcanic eruptions, and lightening strikes. Man made by fusing sand with soda, lime, and sometimes other ingredients and cooling rapidly.
Melanophlogite is a rare silicate mineral and a polymorph of silica (SiO2) that forms around volcano vents. It has a zeolite-like porous structure which results in relatively low and not well-defined values of its density and refractive index.
Coesite is a form (polymorph) of silicon dioxide SiO2 that is formed when very high pressure (2–3 gigapascals), and moderately high temperature (700 °C, 1,300 °F), are applied to quartz.
Stishovite is found at sites of meteorite impact, formed by shock metamorphism of quartz at temperatures 1200 deg C and pressures »100 kbar.
Silica Gel
Opal-A
Opal-CT
Opal-C
Micro-Quartz
Flamboyant Quartz
Silica melt
Silica Gas
Cooling SiO2 back down again.

Water has exactly the opposite charge, and the two of them inter-react in many delightful ways.

A word about electricity and magnetism. They are opposite sides of the same coin. You can't have one without the other.
Electro-Magnetism! - - - - - - - - - - -

As the most common mineral in earth's crust, it usually combines with other SiO2 molecules to form SiO4 tetrahedrons, which then join other tetrahedrons - becoming ever larger Givers and Takers!.


ALL substances go through a series of changes with variations of heat and pressure.

Phase Change Diagrams help us visualize the processes. The "Triple Point" is where solid, liquid, and gas all co-exist.
The "Critical Point" is where mater changes to a plasma.

Here is a diagram for SiO2 -

The letters pH stand for potential of Hydrogen. The pH scale was devised in 1923 by Danish biochemist Søren Peter Lauritz Sørensen (1868-1969). In effect pH is a measure of the concentration of hydrogen ions (that is, protons) in a substance.
pH is a measure of how acidic/basic water is. The range goes from 0 - 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base. Again, pH is just a measure of the relative amount of free hydrogen and hydroxyl ions in the water.

What does this have to do with silica? A LOT!

Some salts dissolve easier in acids, others in bases.
1. An acid is a compound of Hydrogen (or hydroxyl) and a Nonmetallic Element.
Acids turn blue litmus paper red
2. A Base is a Metallic Oxide with water.
Bases turn red litmus paper blue.
3. A Salt is formed when a Base neutralizes an Acid.
A Salt can be described as a compound where hydrogen atoms have been replaced with metallic elements.
Acids turn blue litmus paper red
2. A Base is a Metallic Oxide with water.
Bases turn red litmus paper blue.
3. A Salt is formed when a Base neutralizes an Acid.
A Salt can be described as a compound where hydrogen atoms have been replaced with metallic elements.
Low Temperature SiO2 forms:
Silica Jells are amorphous and porous forms of silicon dioxide, consisting of an irregular tridimensional framework of alternating silicon and oxygen atoms with nanometer-scale voids and pores. The voids may contain water or some other liquids, or may be filled by gas or vacuum.
Opal-A has a solubility of 120–140 ppm in normal marine sediment pore water, cristobalite25–30 ppm, and quartz 6–10 ppm. The dissolution of opal-A generally results in supersaturation with respect to opal-CT, which is precipitated in preference to quartz.
Opal-CT consists of small bladed crystals that form spherical lepispheres 5–10mm in diameter. (Lepisphere : A microcrystalline, bladeshaped crystal of a metastable variety of quartz, composed of cristobalite with interlayered lattices of tridymite, aggregates of which often occur during the transformation of opal into quartz chert.)
Opal-C consists of small bladed cristobalite crystals that form spherical lepispheres 5–10mm in diameter.
Quartz The solubility in water of both amorphous silica and quartz increases rapidly above pH 9, and thus a sharp reduction in alkalinity from pH>9 results in silica precipitation. Precipitation generally takes place from dilute solutions over longer periods of time. The precipitation of silica, particularly around hot springs, occurs due to the evaporation of silica-saturated water. Silica solubility is higher in hot water, and thus cooling is a trigger for silica deposition. Boiling is also a trigger for silica precipitation in hydrothermal systems.
VERY HIGH TEMPERATURE FORMS:
Glass is a non-crystalline, often transparent amorphous solid typically transparent or translucent, created by meteor impacts, volcanic eruptions, and lightening strikes. Man made by fusing sand with soda, lime, and sometimes other ingredients and cooling rapidly.
Melanophlogite is a rare silicate mineral and a polymorph of silica (SiO2) that forms around volcano vents. It has a zeolite-like porous structure which results in relatively low and not well-defined values of its density and refractive index.
VERY HIGH PRESSURE FORMS:
Coesite is a form (polymorph) of silicon dioxide SiO2 that is formed when very high pressure (2–3 gigapascals), and moderately high temperature (700 °C, 1,300 °F), are applied to quartz.
Stishovite is found at sites of meteorite impact, formed by shock metamorphism of quartz at temperatures 1200 deg C and pressures »100 kbar.
We have a lot to cover and need a "Topic transporter" to get us through it all.
Silica Gel
Opal-A
Opal-CT
Opal-C
Micro-Quartz
Flamboyant Quartz
Silica melt
Silica Gas
Cooling SiO2 back down again.