Post by 1dave on Nov 22, 2020 11:34:43 GMT -7
I wrote much of this long ago, BUT our local Rock Club website has gone the way of websites past, So I'll try to re-construct it from old notes.
Introduction to Rocks
How could anyone be interested in rocks? After all it is just the stuff our bodies are composed of, that surrounds us, provides our food shelter and clothing, and a few baubles like silver, gold, platinum, diamonds, rubies, emeralds and other gems.
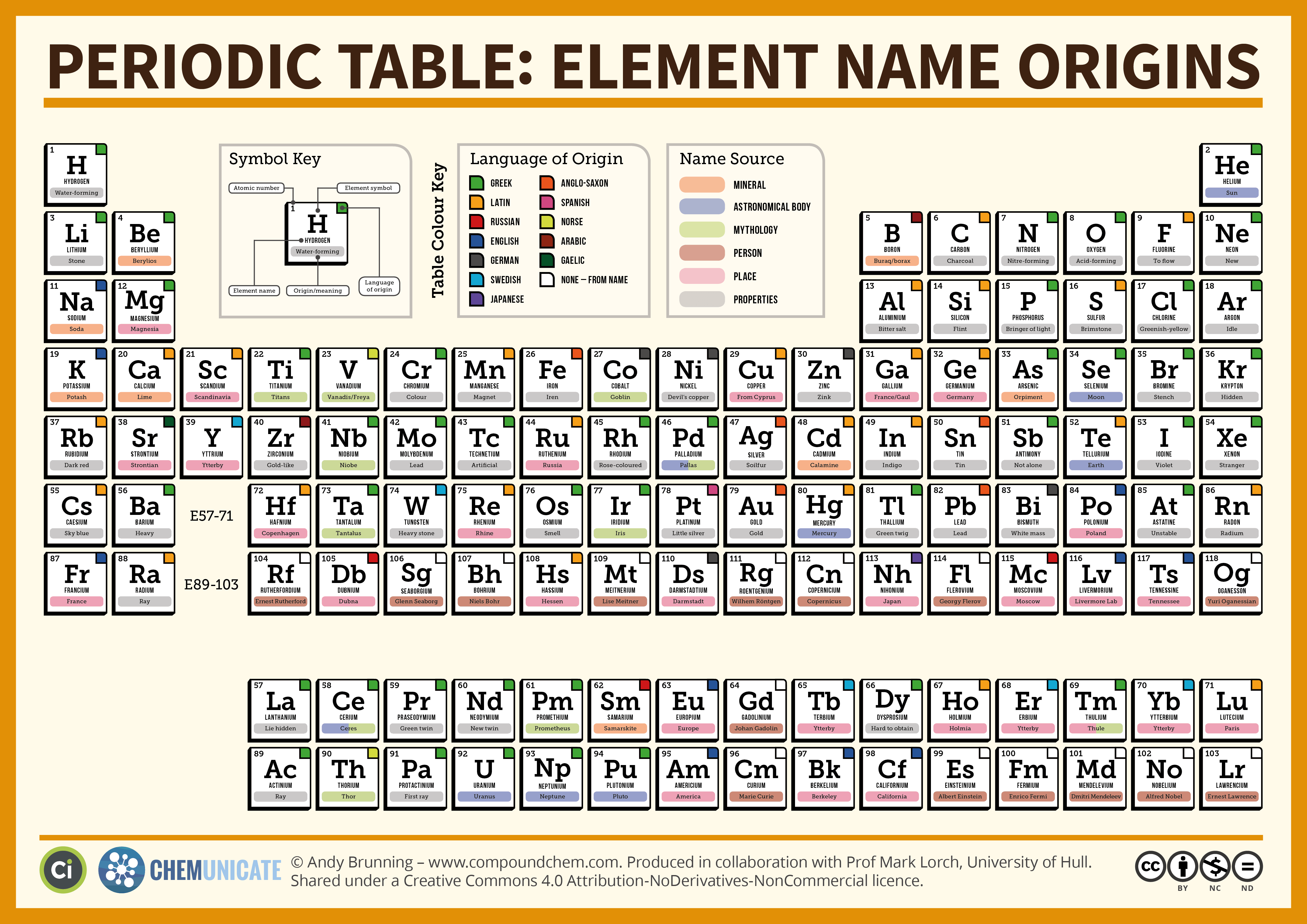
One glance at the Periodic Table of the Elements can be intimidating, but most of those elements are so scarce that you will seldom encounter them in nature.
In spite of 13.7 billion years of stars busily converting hydrogen into ever heavier elements, 97% of all interstellar space is still only Hydrogen.
"Only Hydrogen"? That is the building block from which ALL of creation has been assembled! When God said "Let there be light," much of the light was furnished by hydrogen!

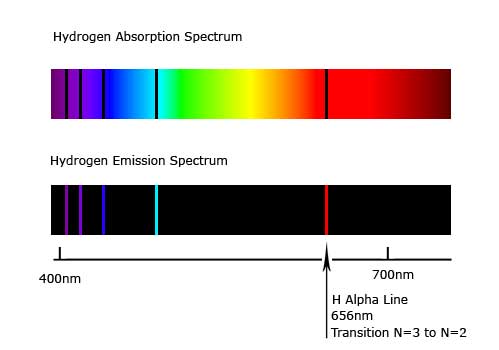
As energy increases or decreases, each element changes the light involved, for each element has a different dimension.

Here in the Milky Way Galaxy Supernovas have changed what elements are near us.




Our oceans contain traces of every element, but most of it is Hydrogen and Oxygen.

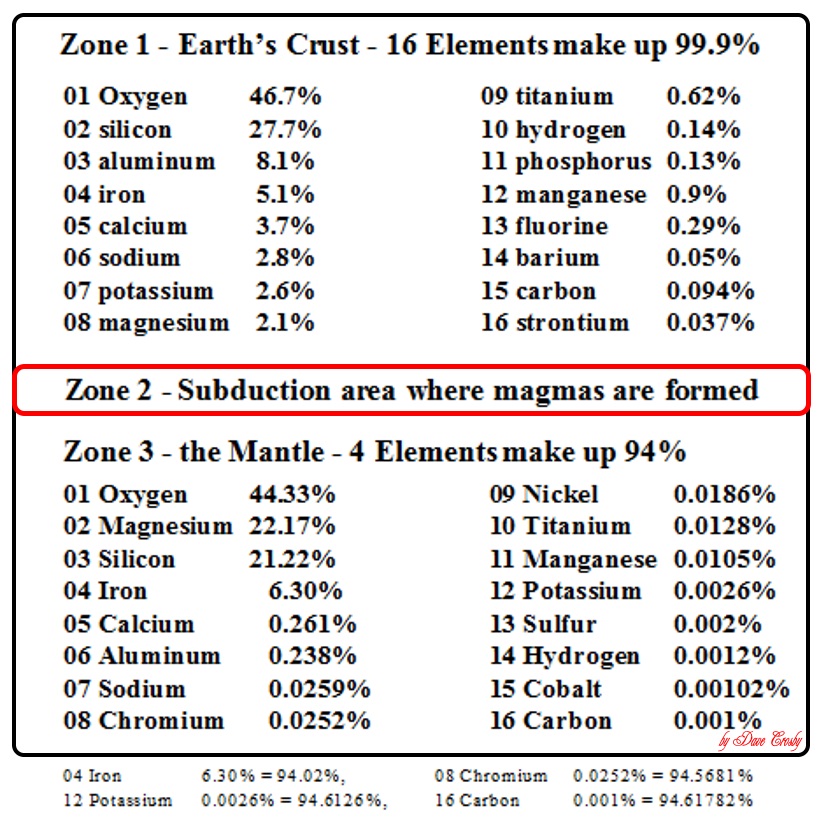
By far most of the rocks you see on land are constructed of only 8 elements:
Oxygen, Silicon, Aluminum, Sodium, Potassium, Calcium, Iron, and Magnesium.
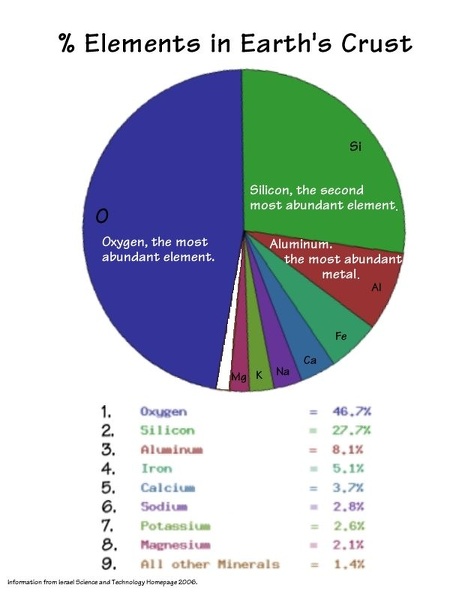
(Surprise - the 9th is titanium!)
Not only do they form the most commonly generated and stable elements, they are also the lightest, so they tend to float to the tops of continents.
Igneous rocks come out of volcanoes and rifts. Some heavy elements tend to crystallize at high temperatures and drop to the bottom of the magma chambers, changing the composition of the remaining magma over time.
Sedimentary rocks are the remnants of old magmas that have cooled, weathered and broken down over time to become valley, beach, lake and ocean bottom fill.
Metamorphic rocks result from Igneous, Sedimentary, and/or previous metamorphic materials being deeply buried and transformed by heat and pressure or other chemical agents.
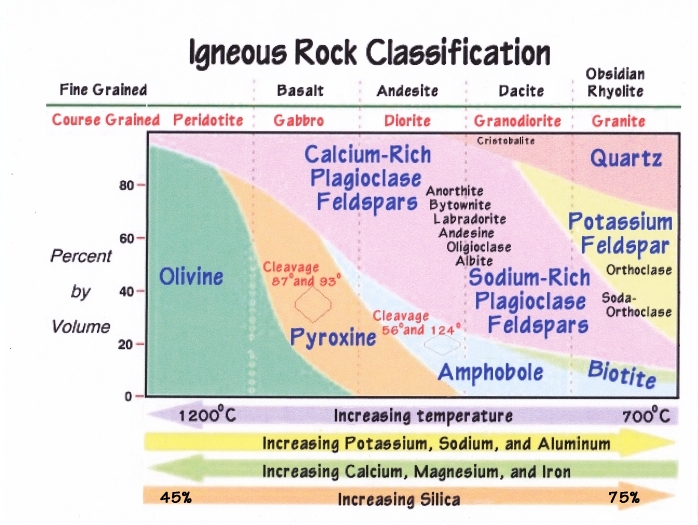
There is far more Oxygen and Silica than anything else on the surface of the earth. Silicon Dioxide is found almost everywhere, and in almost everything.
Igneous: SiO2 is polymorphous. That is it takes many forms depending on temperature and/or pressure. High and Low Quartz, Cristobalite, Tridymite, Coesite, Keatite, Shistovite.
Sedimentary: Boulders, Rocks, Gravel, Sand are mostly size descriptions, but most of it is SiO2.
SiO2 is hard to dissolve UNLESS there is hot water AND alkali around. Then it easily goes into solution. It then percolates through the surrounding area and ACID precipitates it in decomposing buried trees, bones, or other voids as Agate, Chalcedony, and Opal.
Metamorphic: Sedimentary rocks high in SiO2 are compressed into Quartzite.
Igneous: “Feldspars” (Field Crystals) are the most common of all igneous and metamorphic rocks on Earth, but the combinations of the six other main elements with SiO2 presents a wide variety of guises. The various elements are so interchangeable they present many “Solid Series Solutions” varying from 100% of one element to 100% of another.
Often a sheet of one variety of feldspar will be followed by a sheet of another producing diffraction gratings that creates attractive schillers seen as sunstone, moonstone, bronzestone, and the highly prized labradorite.
Sedimentary: The feldspars break down below sand into the finest of sediments known as clay, of which there are many varieties: Argil -potter’s clay, Kaolin -ceramic clay, etcetera.
Volcanic ash sometimes thousands of feet thick blanketed large portions of the earth, then altered into Bentonite Clay that swells when it gets wet.
Clay that swells when it gets wet.
One such field at the end of the Triassic (206 million years ago) buried trees all around the Four Corners region. It is now known as the Chinle Formation, or the Painted Desert. It is filled with petrified wood that is world renowned for its beauty.
Another field at the end of the Jurassic (144 m.y.a.) is known as the Morrison Formation. It is filled with petrified dinosaur bones.
Metamorphic: Gneiss, Schist, Slate, Marble, and a unique group of new minerals:
corundum, fluorite, garnet, graphite, kyanite, mica, wollastonite, pyroxene, tremolite, spinel, starurolite, talc, topaz, tourmaline, and zoisite.
Impact
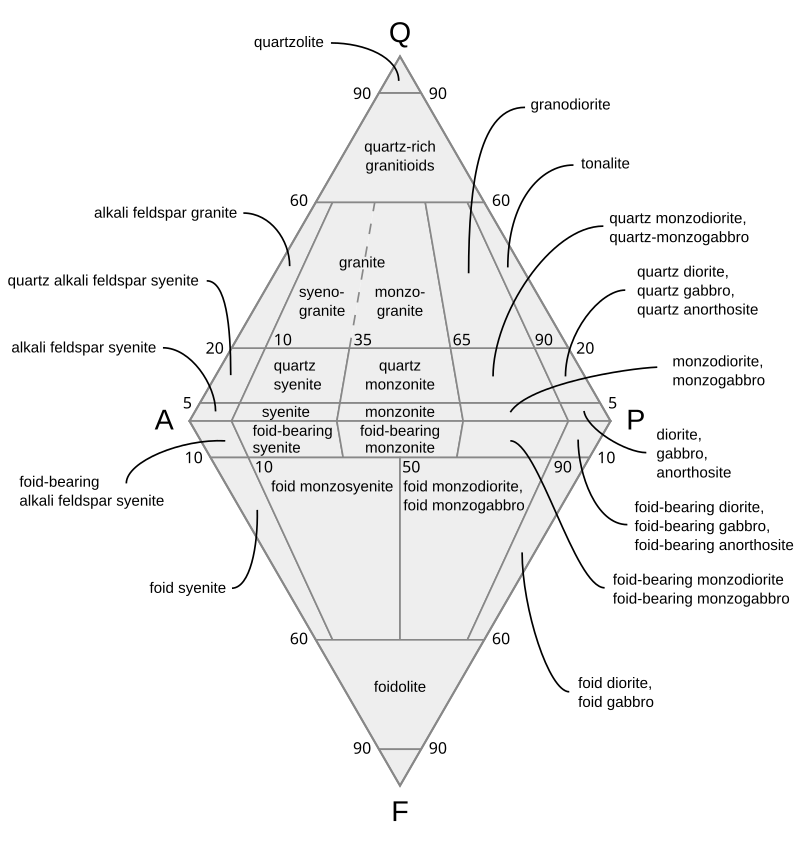
Granite is an igneous rock composed of Quartz, Feldspar, and Mica crystals. But, which Feldspar? How much Quartz? What kind of mica? Obviously there are many kinds of Granite.
Rhyolite is granite that reached the surface before cooling.
If it cooled quickly it became Obsidian.
If it also had over 2% water in it, it became Perlite.
Micas are Muscovite -pale silver gray, Biotite -brown to black, and Phlogopite -bronze.
The only free iron I know of on earth is in Very Ancient Layers (formed before an oxygen atmosphere was formed) like Ayer’s Rock A.K.A. Uluru in Australia.
Iron reacts easily with oxygen or sulphur to form many common minerals.
Black is either Magnetite Fe3O4 or Goethite Fe2O3.H2O, which is not magnetic.
Red is Hematite Fe2O3.
Brown to Yellow is Limonite Fe2O3.3H2O A.K.A. Rust.
Ilmenite FeTiO3 is gray to black and is common as grains in igneous and metamorphic rocks.
Pale brass yellow in cubes or octahedrons is Pyrite FeS2.
Pale bronze yellow in tablets, radiating or globular is Marcasite FeS2.
These minerals are also the most commonly found minerals in meteorites.
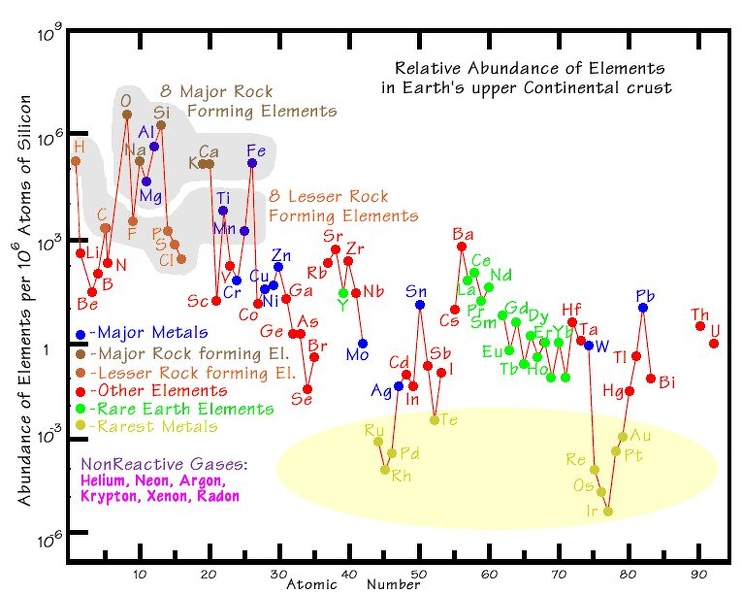
As you can see in the attached chart, the least common of all elements is Iridium. That is why the discovery of a world wide layer of it at the K/T boundary was such a surprise.
Introduction to Rocks
How could anyone be interested in rocks? After all it is just the stuff our bodies are composed of, that surrounds us, provides our food shelter and clothing, and a few baubles like silver, gold, platinum, diamonds, rubies, emeralds and other gems.

One glance at the Periodic Table of the Elements can be intimidating, but most of those elements are so scarce that you will seldom encounter them in nature.
In spite of 13.7 billion years of stars busily converting hydrogen into ever heavier elements, 97% of all interstellar space is still only Hydrogen.
"Only Hydrogen"? That is the building block from which ALL of creation has been assembled! When God said "Let there be light," much of the light was furnished by hydrogen!


As energy increases or decreases, each element changes the light involved, for each element has a different dimension.

Here in the Milky Way Galaxy Supernovas have changed what elements are near us.




Our oceans contain traces of every element, but most of it is Hydrogen and Oxygen.

By far most of the rocks you see on land are constructed of only 8 elements:
Oxygen, Silicon, Aluminum, Sodium, Potassium, Calcium, Iron, and Magnesium.

(Surprise - the 9th is titanium!)
Not only do they form the most commonly generated and stable elements, they are also the lightest, so they tend to float to the tops of continents.

Igneous rocks come out of volcanoes and rifts. Some heavy elements tend to crystallize at high temperatures and drop to the bottom of the magma chambers, changing the composition of the remaining magma over time.
Sedimentary rocks are the remnants of old magmas that have cooled, weathered and broken down over time to become valley, beach, lake and ocean bottom fill.
Metamorphic rocks result from Igneous, Sedimentary, and/or previous metamorphic materials being deeply buried and transformed by heat and pressure or other chemical agents.
There is far more Oxygen and Silica than anything else on the surface of the earth. Silicon Dioxide is found almost everywhere, and in almost everything.
Igneous: SiO2 is polymorphous. That is it takes many forms depending on temperature and/or pressure. High and Low Quartz, Cristobalite, Tridymite, Coesite, Keatite, Shistovite.
Sedimentary: Boulders, Rocks, Gravel, Sand are mostly size descriptions, but most of it is SiO2.
SiO2 is hard to dissolve UNLESS there is hot water AND alkali around. Then it easily goes into solution. It then percolates through the surrounding area and ACID precipitates it in decomposing buried trees, bones, or other voids as Agate, Chalcedony, and Opal.
Metamorphic: Sedimentary rocks high in SiO2 are compressed into Quartzite.
Igneous: “Feldspars” (Field Crystals) are the most common of all igneous and metamorphic rocks on Earth, but the combinations of the six other main elements with SiO2 presents a wide variety of guises. The various elements are so interchangeable they present many “Solid Series Solutions” varying from 100% of one element to 100% of another.
Often a sheet of one variety of feldspar will be followed by a sheet of another producing diffraction gratings that creates attractive schillers seen as sunstone, moonstone, bronzestone, and the highly prized labradorite.
Sedimentary: The feldspars break down below sand into the finest of sediments known as clay, of which there are many varieties: Argil -potter’s clay, Kaolin -ceramic clay, etcetera.
Volcanic ash sometimes thousands of feet thick blanketed large portions of the earth, then altered into Bentonite
 Clay that swells when it gets wet.
Clay that swells when it gets wet.One such field at the end of the Triassic (206 million years ago) buried trees all around the Four Corners region. It is now known as the Chinle Formation, or the Painted Desert. It is filled with petrified wood that is world renowned for its beauty.
Another field at the end of the Jurassic (144 m.y.a.) is known as the Morrison Formation. It is filled with petrified dinosaur bones.
Metamorphic: Gneiss, Schist, Slate, Marble, and a unique group of new minerals:
corundum, fluorite, garnet, graphite, kyanite, mica, wollastonite, pyroxene, tremolite, spinel, starurolite, talc, topaz, tourmaline, and zoisite.
Impact
Granite is an igneous rock composed of Quartz, Feldspar, and Mica crystals. But, which Feldspar? How much Quartz? What kind of mica? Obviously there are many kinds of Granite.
Rhyolite is granite that reached the surface before cooling.
If it cooled quickly it became Obsidian.
If it also had over 2% water in it, it became Perlite.
Micas are Muscovite -pale silver gray, Biotite -brown to black, and Phlogopite -bronze.
The only free iron I know of on earth is in Very Ancient Layers (formed before an oxygen atmosphere was formed) like Ayer’s Rock A.K.A. Uluru in Australia.
Iron reacts easily with oxygen or sulphur to form many common minerals.
Black is either Magnetite Fe3O4 or Goethite Fe2O3.H2O, which is not magnetic.
Red is Hematite Fe2O3.
Brown to Yellow is Limonite Fe2O3.3H2O A.K.A. Rust.
Ilmenite FeTiO3 is gray to black and is common as grains in igneous and metamorphic rocks.
Pale brass yellow in cubes or octahedrons is Pyrite FeS2.
Pale bronze yellow in tablets, radiating or globular is Marcasite FeS2.
These minerals are also the most commonly found minerals in meteorites.
As you can see in the attached chart, the least common of all elements is Iridium. That is why the discovery of a world wide layer of it at the K/T boundary was such a surprise.