Post by 1dave on Dec 28, 2020 11:54:36 GMT -7
A lithophysa (plural lithophysae, from Greek lithos "stone" + physan "to blow")[1] is a felsic volcanic rock with a spherulitic structure and interior cavity with concentric chambers. Its outer shape is spherical or lenticular. They vary in size from very small up to twelve feet in diameter depending on the age of the magma chamber. These rocks are usually found within obsidian or rhyolite lava flows. Magmas low in feldspar minerals may produce a version known as "snowflake obsidian.'[2]
These cavities are believed to be caused by expanding gases in tuffs and rhyolitic lavas before solidification.[1] If it has become lined with crystals it may be referred to as a geode or filled partially or fully with agate, jasper or opal is a thunderegg. The term vug is also used for similar cavities although the meaning of vug is usually restricted to cavities in rocks formed by the removal of material such as soluble minerals. These cavities usually contain layers of various colors (red, pink, gray, etc.) composed by crystals of quartz,[1] chalcedony,[3] hematite, fluorite[4] and various colored oxides or other minerals.[1]
A collection of lithophysae "thundereggs."
Lithophysae are one of the many forms of silica (SiO2),[3] as quartz,[1] agate,[1] opal,[1] chalcedony, etc. They can have an average diameter 5–20 centimetres (2.0–7.9 in), with a maximum of 60–80 centimetres (24–31 in).[3] Lithophysae are also related to the spherulites found in obsidians on the Italian island of Lipari, in Yellowstone National Park, and other places.[5]
Basics are that Si-O2 structures change with temperature and pressure. (AND the presence of other elements)
Heating up from Zero to 10,000, first the core of one element will increase vibrating to the point where the "Forbidden Zone" will increase to the point where the electrons are forced to move out to the next path; then the other will soon follow suit. this increases their sizes
- Quantum Mechanics -.
This is a predictable and repeatable path, occurring at precise temperatures and pressures.
Increasing from room temperature: SiO2 gel > opal > needles > spheroids > chalcedony > moganite > alpha quartz > beta quartz > 7 forms of tridymite > 2 forms of cristobalite > spheroids > liquid > gas > plasma.
Cooling Down is a different matter.
External Structure tends to hold on to shape, sometimes obvious as "crazing" as b quartz cools below 575oC into a quartz.
Most volcanoes are subduction volcanoes with a life span of only several thousand years. Not time enough to grow very large spheroids.
2. Spheroids (Lithophasae) in Rhyolite and Obsidian
The mineral content of magma is constantly changing as iron and magnesium combine to form olivine crystals; calcium, sodium, and potassium form feldspars and they drop out of solution, making it more and more silica laden over time.
- - - - - - - - - - - - - - - - - - - -
At 1,100oC crystobalite needles form, making "puffballs" with very hot water trapped between the needles. The needles push out the calcium, sodium, and potassium molecules. All that movement causes friction and makes the magma hotter. It becomes too hot for crystobalite to form, but just right for Perthite feldspar to form a sphere around the puffball. The magma cools and crystobalite needles resume.
You see them all the time as "Snowflake Obsidian."



A Thunderegg Siamese Twin that formed in obsidian.

As the flow slowly cools and contracts, pressure is released and the water is finally allowed to expand into steam, forcing the needles apart.

As the gas forces the matrix apart around the thunderegg, "sutures" are formed where the sections break apart.
If the gas escapes the sutures are sucked in.

Thunderegg Cores - the outer shells removed.


I've become a forgetful old man! This is another thread I started and never got back to.
I'm tempted to start a new thread " The Yin and Yang of SiO2 "
Down near absolute ZERO all is quiet on the western front.
Silica and oxygen bonds are at their shortest lengths. all the groups are similar shaped tetrahedrons that stack together in regular fashion to make identical crystals.

As temperatures increase, just like molting creatures changing their exoskeletons, suddenly all the oxygen atoms get bigger ad their bonds jump to a new length! The crystals have to change shapes, just like a brick wall when you change the brick shapes.
Later, the bigger and heavier silicon atoms follow suit, and again, the crystal shapes must change!
Shapes change from a to b, then c, d, e, f ,g, h, but then begin repeating themselves again, only in a larger size as A, B, C, D, E, . . .
For 50 years I studied the high temperature forms of quartz a and b, tridymite a and b, cristobalite a and b.
Now I am realizing those same shapes occurred at and below room temperature as silicic acid polymorphs as monomers, dimers, trimers, etc.!



These cavities are believed to be caused by expanding gases in tuffs and rhyolitic lavas before solidification.[1] If it has become lined with crystals it may be referred to as a geode or filled partially or fully with agate, jasper or opal is a thunderegg. The term vug is also used for similar cavities although the meaning of vug is usually restricted to cavities in rocks formed by the removal of material such as soluble minerals. These cavities usually contain layers of various colors (red, pink, gray, etc.) composed by crystals of quartz,[1] chalcedony,[3] hematite, fluorite[4] and various colored oxides or other minerals.[1]
A collection of lithophysae "thundereggs."
Lithophysae are one of the many forms of silica (SiO2),[3] as quartz,[1] agate,[1] opal,[1] chalcedony, etc. They can have an average diameter 5–20 centimetres (2.0–7.9 in), with a maximum of 60–80 centimetres (24–31 in).[3] Lithophysae are also related to the spherulites found in obsidians on the Italian island of Lipari, in Yellowstone National Park, and other places.[5]
Basics are that Si-O2 structures change with temperature and pressure. (AND the presence of other elements)
Heating up from Zero to 10,000, first the core of one element will increase vibrating to the point where the "Forbidden Zone" will increase to the point where the electrons are forced to move out to the next path; then the other will soon follow suit. this increases their sizes
- Quantum Mechanics -.
This is a predictable and repeatable path, occurring at precise temperatures and pressures.
Increasing from room temperature: SiO2 gel > opal > needles > spheroids > chalcedony > moganite > alpha quartz > beta quartz > 7 forms of tridymite > 2 forms of cristobalite > spheroids > liquid > gas > plasma.
Cooling Down is a different matter.
External Structure tends to hold on to shape, sometimes obvious as "crazing" as b quartz cools below 575oC into a quartz.
Most volcanoes are subduction volcanoes with a life span of only several thousand years. Not time enough to grow very large spheroids.
2. Spheroids (Lithophasae) in Rhyolite and Obsidian

The mineral content of magma is constantly changing as iron and magnesium combine to form olivine crystals; calcium, sodium, and potassium form feldspars and they drop out of solution, making it more and more silica laden over time.
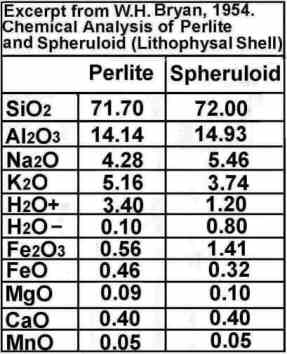
Basalt ~ 45% Silica, Andesite ~ 55% Silica, Rhyolite ~ 65% Silica
BUT the Thunderegg Shells reach over 70% Silica!
BUT the Thunderegg Shells reach over 70% Silica!
- - - - - - - - - - - - - - - - - - - -

At 1,100oC crystobalite needles form, making "puffballs" with very hot water trapped between the needles. The needles push out the calcium, sodium, and potassium molecules. All that movement causes friction and makes the magma hotter. It becomes too hot for crystobalite to form, but just right for Perthite feldspar to form a sphere around the puffball. The magma cools and crystobalite needles resume.
You see them all the time as "Snowflake Obsidian."



A Thunderegg Siamese Twin that formed in obsidian.

As the flow slowly cools and contracts, pressure is released and the water is finally allowed to expand into steam, forcing the needles apart.

As the gas forces the matrix apart around the thunderegg, "sutures" are formed where the sections break apart.
If the gas escapes the sutures are sucked in.

Later the Spheroid voids are filled with agate the same way as they are in basalt voids.
Finally the temperature becomes so cool crystobalite needles can no longer exist and they revert to other SiO2 forms, but they sometimes leave imprints to prove they once existed.
Finally the temperature becomes so cool crystobalite needles can no longer exist and they revert to other SiO2 forms, but they sometimes leave imprints to prove they once existed.
Thunderegg Cores - the outer shells removed.


I've become a forgetful old man! This is another thread I started and never got back to.
There is so much to learn!
I'm tempted to start a new thread " The Yin and Yang of SiO2 "
Down near absolute ZERO all is quiet on the western front.
Silica and oxygen bonds are at their shortest lengths. all the groups are similar shaped tetrahedrons that stack together in regular fashion to make identical crystals.

As temperatures increase, just like molting creatures changing their exoskeletons, suddenly all the oxygen atoms get bigger ad their bonds jump to a new length! The crystals have to change shapes, just like a brick wall when you change the brick shapes.
Later, the bigger and heavier silicon atoms follow suit, and again, the crystal shapes must change!
Shapes change from a to b, then c, d, e, f ,g, h, but then begin repeating themselves again, only in a larger size as A, B, C, D, E, . . .
For 50 years I studied the high temperature forms of quartz a and b, tridymite a and b, cristobalite a and b.
Now I am realizing those same shapes occurred at and below room temperature as silicic acid polymorphs as monomers, dimers, trimers, etc.!