Post by Admin on Aug 15, 2021 11:20:42 GMT -7

A hydrogen atom is about 0.1 nanometers, and a cesium atom is around 0.3nm.
the electrons are traveling around them at about 2,200 kilometers per second.
Q: Are they like the moon, always facing the proton; spinning clockwise or counter clockwise, or always facing the same direction?
We talk about clockwise VS counter clockwise as bring opposites.
Actually they are just opposite sides of the same thing.

Looking at the Hydrogen Arom and electron from a different mind set -


Electrons are spinning magnets.
Two can only get along when they are upside down to each other so they mesh like gears.


In orbits they have to line up in order.

Naturally the same thing has to happen in magnets.

We have discovered that at different energy levels, atoms emit increasingly higher electromagnetic waves.
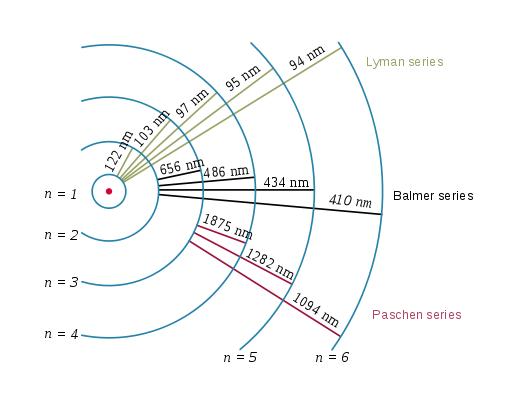
What is happening? The electron is forced to spend more time in ever increasing spiral paths!
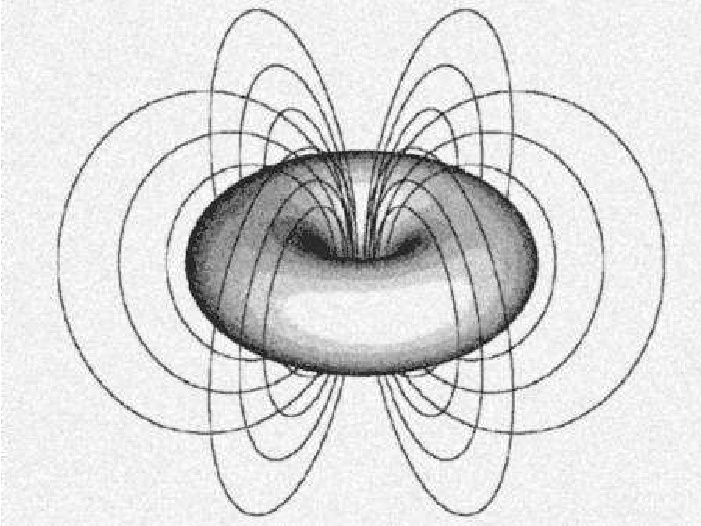

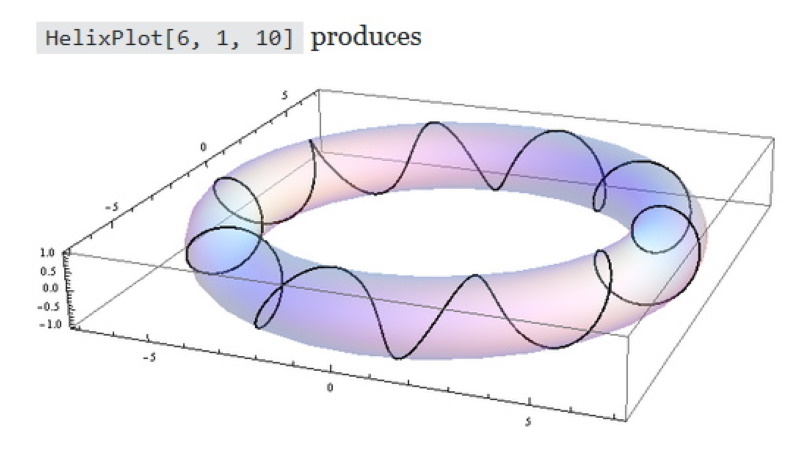
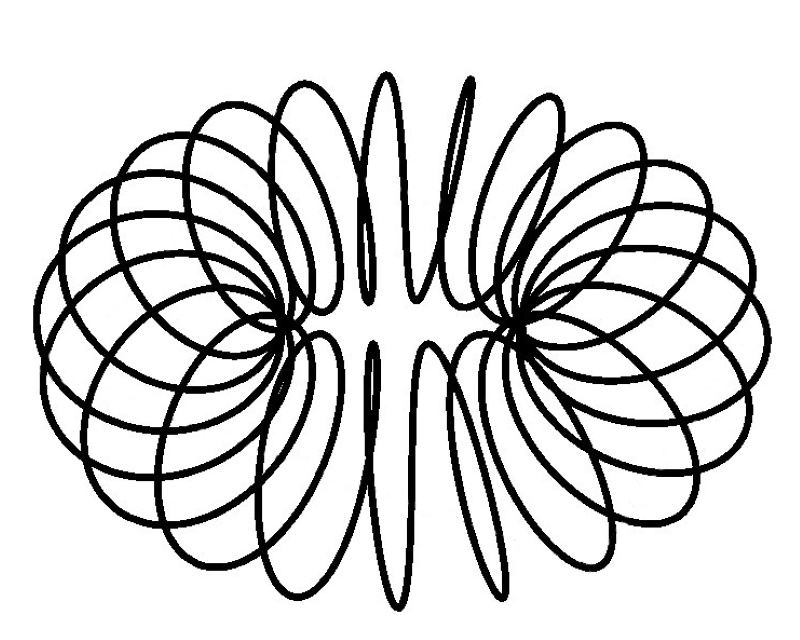
The electron is in all of these areas some of the time, Energy levels determine where it is most of the time.
It is moving so fast in such small confined areas that the spacing is created by self repulsion!

The electron IS the magnetic field!
Augmented or weakened by the electron orbist around it.
Augmented or weakened by the electron orbist around it.
The collision of two hydrogen atoms spinning clockwise bounce them far apart, BUT if spinning opposite, they mesh like gears AND cancel out the magnetic field around them!

When 4 hydrogen atoms are star crushed into 1 helium atom, the two electrons mesh in the same way. That is why helium -and all the "Nobel Gases" are electromagnetically neutral.

But IRON is a different matter!

When a current flows through iron, the molecules flip so they are all pointing the same direction and the magnetic field is enhanced and can become "permanent."
With electricity constantly flowing just like in an electromagnet!

But think about levers and gears inside molecules!

Electron Spin
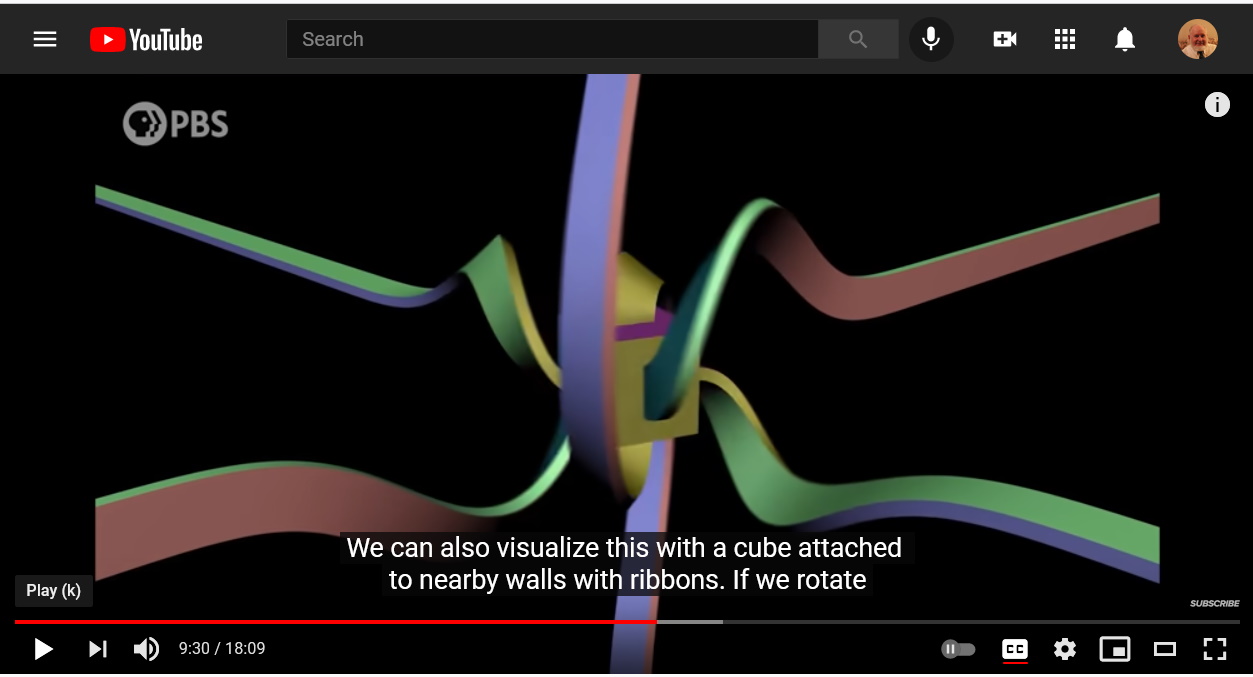
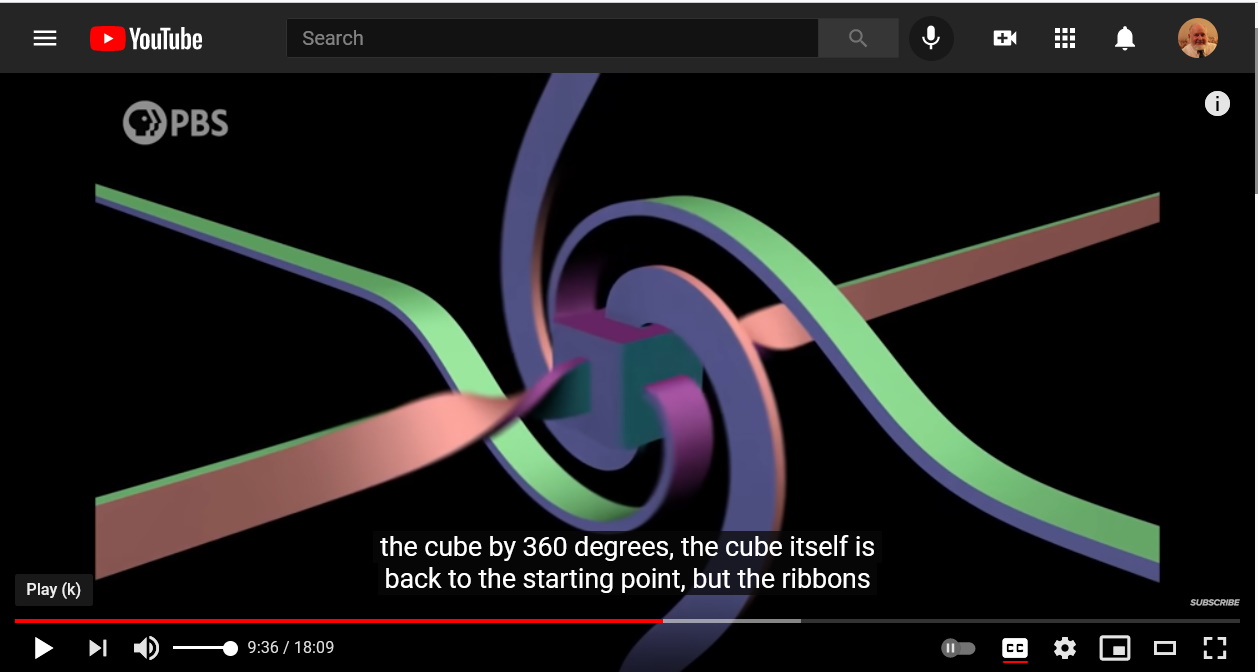
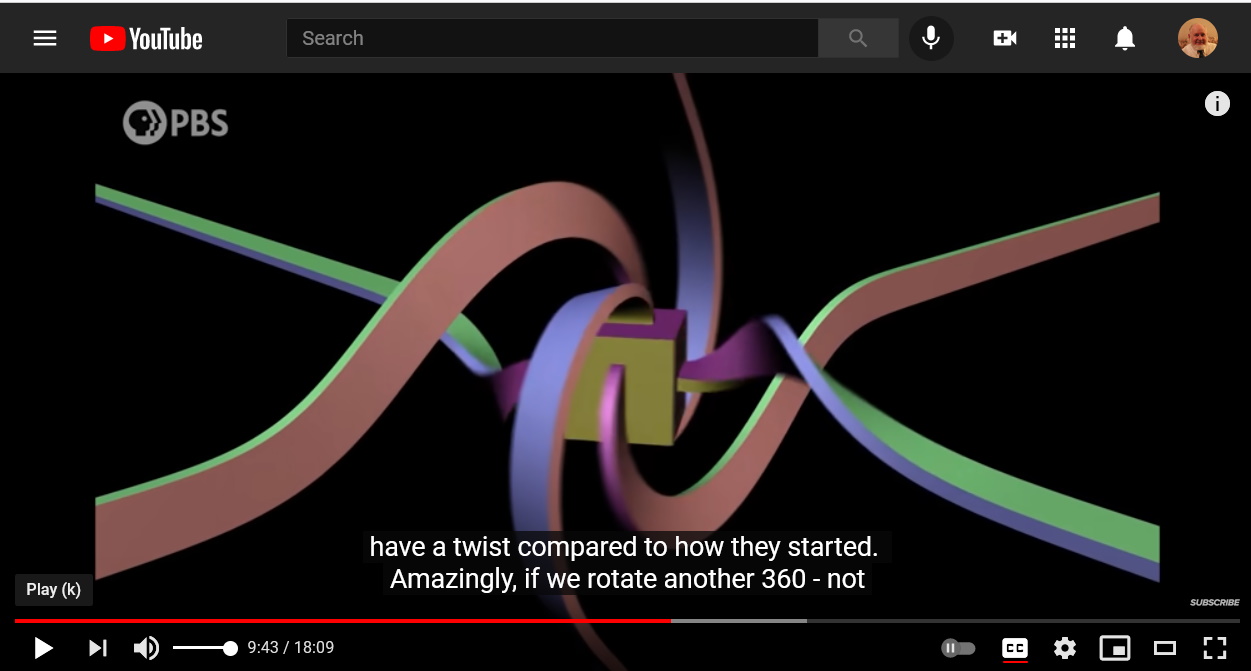

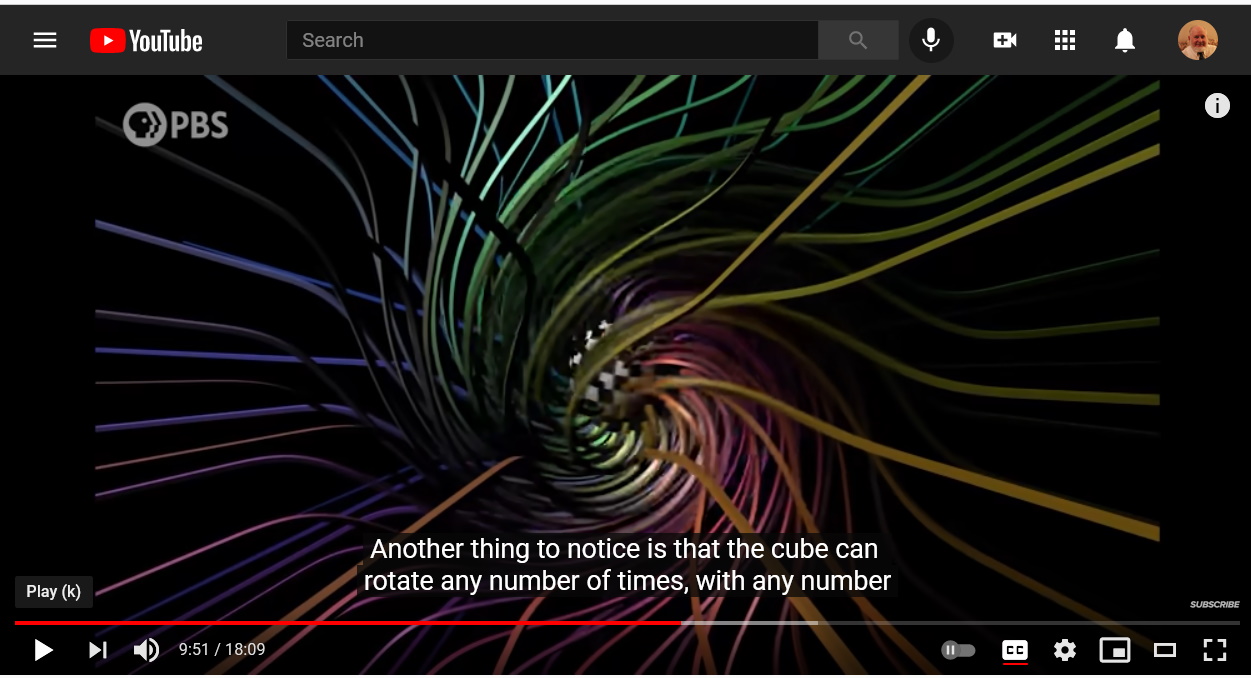


Electric charges are sources of electric fields.
Magnetic charges are sources of magnetic fields.
An electric field exerts a force on an electric charge, which is proportional to the magnitude of both the field and the charge, and parallel to the field.
A magnetic field exerts a force on a magnetic charge, which is proportional to the magnitude of both the field and the charge, and parallel to the field.
Therefore, opposite electric charges attract and like electric charges repel, and likewise opposite magnetic charges attract and like magnetic charges repel.
Moving electric charges generate magnetic fields.
Moving magnetic charges generate electric fields.
Magnetic fields exert forces on electric charges that are perpendicular to both the field and the charge's velocity, and proportional to the magnitude of both the field and the charge.
Electric fields exert forces on magnetic charges that are perpendicular to both the field and the charge's velocity, and proportional to the magnitude of both the field and the charge.
Therefore, parallel electric currents attract and antiparallel electric currents repel, and likewise parallel magnetic currents attract and antiparallel magnetic currents repel.
The energy stored in the electric field is proportional to the square of the field's magnitude.
The energy stored in the magnetic field is proportional to the square of the field's magnitude.
A changing electric field is accompanied by a magnetic field.
A changing magnetic field is accompanied by an electric field.
All the above facts can be traced to the fact that the hypothetical Maxwell's equations in the presence of magnetic charges:
∇⋅E∇⋅B∇×E∇×B=kρe=kρm=−1c∂B∂t−kcJm=kcJe+1c∂E∂t
combined with the Lorentz force law in the presence of magnetic charges:F=qe(E+vc×B)+qm(B−vc×E)
are invariant under a duality transformation of the following form:
q eq mE B ←qecosθ+qmsinθ←qmcosθ−qesinθ←Ecosθ+Bsinθ←Bcosθ−Esinθ
(Note: Since SI and cgs-Gaussian units both suck, I am using a hybrid unit system, which is described here
. The magnetic field is scaled up by a factor of c, and k is 4π
times Coulomb's constant.)
In other words, the electromagnetic charge of a particle can be thought of as a point on an E-M plane. Purely electrically charged particles lie along the x axis, and purely magnetically charged particles lie along the y axis. (A neutrino, which has no electric or magnetic charge, would lie at the origin.) If we rotated all particles uniformly by some fixed angle, everything would keep on going like it did before. For example, we may choose θ
to be 90 degrees, which would change a particle of E/M charge (qe,qm) to one with E/M charge (qm,−qe)
. In other words it would turn an electric monopole (such as an electron) into a magnetic monopole and vice versa.
So, if somehow all the charged particles in the universe were somehow replaced with magnetic monopoles, we wouldn't be able to notice. So in that deep sense, there is no difference between electric and magnetic charge.
What is interesting is that all particles that are known to humanity seem to lie along a single line through the origin on the E-M plane. We can choose the E axis however we want, and the duality transformations guarantee that the laws of electromagnetism take the same form. For convenience, we choose our E axis along that line. Measured relative to that choice of axes, then, all electromagnetically charged particles we know of are purely electrically charged, and have no magnetic charge. You could just as well choose the M axis to be along that line, though. Or you could choose both the E and M axes to not be along that line, in which case all the charged particles we know of have a combination of electric and magnetic charge (but the ratio is the same for all of them; they all lie along a line in the E-M plane, a fact which is not changed by our choice of axes).
In summary, the difference is purely one of convention. To distinguish between electric and magnetic charge, we define the electron to be electrically charged, not magnetically charged. That's the real answer.
###################################
What are differences and similarities between electric fields and magnetic fields?
Ad by CapitalOne Shopping
Before you shop at Amazon Prime, read this.
The dead giveaway that tells you when Amazon’s giving you a better price than other retailers.
5 Answers
Ramon E Lopez
, Professor of Physics, Heliosphysics researcher
Answered June 29, 2017
Originally Answered: What are the differences between magnetic and electrical field?
Let’s begin briefly with what is a field. A field is a quantity that extends through space, and at each point in space has a magnitude and direction. Fields can change in time and contain energy.
A magnetic field is created by an electric current (moving electric charges) and it exerts a force on a moving charged object in a direction perpendicular to the motion and to the field direction (so a charged object moving parallel to a magnetic field, or not moving at all, experiences no magnetic force).
An electric field is created by electric charges and exerts a force on another electric charge parallel (or anti parallel) to the field direction.
Magnetism in everyday experience is often a force between magnets, or between a magnet and a magnetic material. Those forces operate because on the molecular and atomic scale, there are electrical charges in motion that can create magnetic fields and experience magnetic forces.
Magnetic and electric fields are flip sides of the same coin, united by motion and changes in space and time. This is why we speak of “electromagnetism.” There are four fundamental equations that describe electric and magnetic fields, their sources, and the relationship between them. These are called the Maxwell Equations after the great 19th century physicist James Clerk Maxwell who united the equations (he didn’t discover them all) and showed that one solution was an electromagnetic wave, which is light.
How fast do electrons move?
As fast as you can get them going! Well not quite. One of the facts of life discovered in the 20th century is that the speed of light (300,000 kilometers per second) is the ultimate speed limit. As you add energy to the electron, it will go faster, but as you get it to go close to the speed of light, you find that you have to add even more energy just to bump it a bit faster. For example, with just over 220,000 eV (which stands for a convenient unit of energy called the "electron-volt"), you can get the electron up to 90% of the speed of light. But to get it to 99.9% (just another 9.9 percentage points), you need a total of over 11 million eV! One way of looking at this is that the electron gets "heavier" (more massive) as it goes ever faster. So it's harder to push it faster. At Jefferson Lab, a typical energy for the electrons in the beam is 4 GeV which is 4 billion eV. That means the electron is traveling at 99.9999992% of the speed of light. Close but still not 100%.
You may wonder how fast the electrons are whizzing around in the atoms around you. A good example (and the most simple to calculate) is the hydrogen atom which is in all our water. A calculation shows that the electron is traveling at about 2,200 kilometers per second. That's less than 1% of the speed of light, but it's fast enough to get it around the Earth in just over 18 seconds. Read up on what happens when nothing can go faster than the speed of light.
Author:
Carl Zorn, Detector Scientist (Other answers by Carl Zorn)
As fast as you can get them going! Well not quite. One of the facts of life discovered in the 20th century is that the speed of light (300,000 kilometers per second) is the ultimate speed limit. As you add energy to the electron, it will go faster, but as you get it to go close to the speed of light, you find that you have to add even more energy just to bump it a bit faster. For example, with just over 220,000 eV (which stands for a convenient unit of energy called the "electron-volt"), you can get the electron up to 90% of the speed of light. But to get it to 99.9% (just another 9.9 percentage points), you need a total of over 11 million eV! One way of looking at this is that the electron gets "heavier" (more massive) as it goes ever faster. So it's harder to push it faster. At Jefferson Lab, a typical energy for the electrons in the beam is 4 GeV which is 4 billion eV. That means the electron is traveling at 99.9999992% of the speed of light. Close but still not 100%.
You may wonder how fast the electrons are whizzing around in the atoms around you. A good example (and the most simple to calculate) is the hydrogen atom which is in all our water. A calculation shows that the electron is traveling at about 2,200 kilometers per second. That's less than 1% of the speed of light, but it's fast enough to get it around the Earth in just over 18 seconds. Read up on what happens when nothing can go faster than the speed of light.
Author:
Carl Zorn, Detector Scientist (Other answers by Carl Zorn)